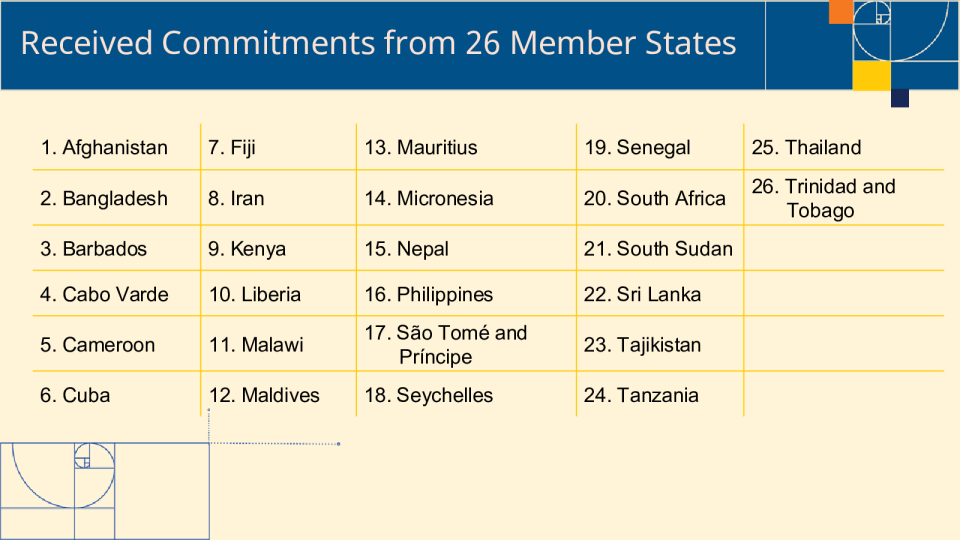
成员国在第二届世界卫生组织传统医学全球首脑会议上承诺的承诺

Afghanistan ✔Establish National Traditional Medicine Policy, Law and Regulation (by 2027)and incorporating WHO Global Traditional Medicine Strategy 2025-2034 into ✔Establish a National TM Research Centre and Dedicated Research-Budget for ✔Establish Health Information System and Integrate Traditional Medicine inGeneral Health Practices Bangladesh ✔Integrate Traditional Medicine in health systems ✔Intellectual Property Protection, Biodiversity & Knowledge Preservation✔Indigenous People’s Rights✔Contribute to Innovation, Scientific Validation, and Market Shaping✔Promote inclusive governance and sustainable✔National Policy as well as National Regularity Authority Barbados ✔Develop Regulations for Herbal Medicine and Good Manufacturing Practices ✔Promote National Traditional Medicine Research as part of the work of theBarbados Living Laboratory (by 2027) ✔Develop and Adopt a National Traditional Medicine Policy (by 2027) ✔Integrate Traditional Medicine in Primary Health Care (by 2028) Cabo Verde ✔Strengthens Financial Protection through TM Integration into National Health ✔Establish GMP-Compliant Production for Herbal Medicines aligned with Safety,Trade, and Economic Growth (by 2026) ✔Establish a National TM Research Centre for Evidence Strengthening (by 2026) ✔Develop and Adopt a National Traditional Medicine (TM) Policy and Law Cameroon ✔Update,harmoniseandoperationalisethe existing National Traditional Medicine Lawand Policy, with clear implementation decrees, governance mechanisms and ✔Strengthen and reposition the IMPM as the national reference institute for traditionalmedicine research, with increased and sustainable funding, and regional researchhubs (by 2026) ✔Pilot the inclusion of selected, evidence-based traditional medicines and practicesinto the Universal Health Coverage scheme, with a focus on indigenous and rural Develop Regulations for Herbal Medicine and Good Manufacturing Practices alignedwith WHO (by 2027) ✔Progressively integrate trained traditional medicine practitioners and validatedtherapies into primary health care (by 2028) Cuba ✔National Program for the Development and Generalization of Natural and ✔Technical Document Summarizing the Country’s Progress by 2026 ✔Technical Document will be Published by the Medical Sciences Publishing House(Editorial Ciencias Médicas in Spanish) Fiji ✔Develop and Adopt a National Traditional Medicine Policy and Program by 2027. ✔Establish a national governance framework in consultation with relevantgovernment and traditional institutions for research and integration of traditional Iran ✔Launch World Health Organization Collaborating Center✔Launch a National Platform for Clinical Trials Kenya ✔Establish a National Policy and Department on Traditional Medicine (by 2028)✔Strengthen Financial Protection through T.M. Integration into National Health Insurance (by 2028) ✔Formalize key documents to promote Traditional Medicine including the NationalCultural Policy on Culture and Heritage, National Environment Action Plan and ✔Legislative safeguards to cement Traditional Medicine including Intellectualproperty rights, equitable benefits and exploitation of communities that own the Liberia ✔Advocate for the passage of the National Public Health Law of Liberia (Drafted2019), which make makes provision for the establishment of the Traditional andComplementary Medicine Board (to be a semi-autonomous entity like the Liberia ✔Strengthen Financial Protection through TM Integration into National Health ✔Establish a National Botanical Garden (Medicinal Plants) for Biodiversity,Conservation, Research, and Local Production (GMP-Compliant Production for ✔Establish a National Digital Database on TCIM Practioners and Services across Malawi ✔National Traditional Medicine Policy Revised by December 2026✔Malawi Traditional Medicine Act Enacted by December 2027✔Malawi Herbal Medicine Regulatory Guidelines Gazetted by December 2026 Maldives ✔Establish National Health Program and Law (by 2027) ✔TM Integration into National Health Insurance (by 2027) ✔Establish GMP-Compliant Production for Herbal Medicines aligned with Safety, Trade, andEconomic Growth (by 2026) ✔Establish a National TM Research Centre for Evidence Strengthening (by 2026) and ✔By 2026: Planning to establish a National Traditional Medicine Committee to providetechnical guidance, coordination, and policy advice, and to explore options for strengtheningresearch and institutional capacity for traditional medicine, including quality assurance ✔By 2027: Planning to develop a national framework for traditional medicine, including policyand legislative considerations, and to explore pathways for the integration of traditional Mauritius ✔Establish National Policy and Office on Traditional Medicine (by 2026) ✔Will incorporate private sector to Strengthen Financial Protection through TM ✔Establish GMP-Compliant Production for Herbal Medicines aligned with Safety,Trade, and Ec